Credit: NASA/GFSCĭuring World War II, Willard Libby – a chemist and graduate of Berkeley – read a paper by W. E. 
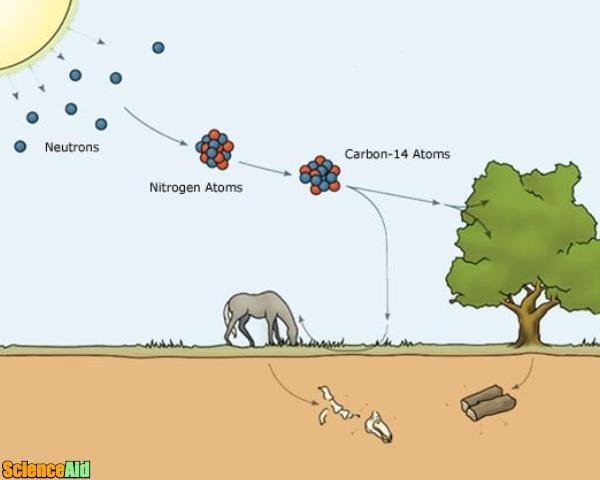
A hydrogen atom is made up of one proton and one electron, but its heavy form, called deuterium, also contains a neutron. This contradicted previous work, which held that it was the product of deuterium (H², or heavy hydrogen) and Carbon 13. At the time, researchers were attempting to determine if any of the elements common to organic matter had isotopes with half-lives long enough to be of value in biomedical research.īy 1940, the half-life of Carbon 14 was determined, as was the mechanism through which it was created (slow neutrons interacting with Nitrogen in the atmosphere). History of Development:Įxperiments that would eventually lead to carbon dating began in the 1939s, thanks to the efforts of the Radiation Laboratory at the University of California, Berkeley. This means that it takes a sample of radiocarbon 5,730 years for half of it to decay back into nitrogen.Īfter about 10 half-lives, the amount of radiocarbon left becomes too minuscule to measure and so this technique isn’t particularly reliable for dating specimens which died more than 60,000 years ago – i.e. In addition, scientists know that the half-life of radiocarbon is 5,730 years. When they die, they cease to consume them, and the isotope of C-14 begins to revert back to its Nitrogen state at an exponential rate due to its radioactive decay.Ĭomparing the remaining C-14 of a sample to that expected from atmospheric C-14 allows the age of the sample to be estimated. Plants and animals absorb both C-12 and C-14 in the course of their natural lifetimes simply by carrying out these basic functions. Radiocarbon enters the biosphere through natural processes like eating and breathing. This property makes it especially useful in a process known as “radiocarbon dating”, or carbon dating for short. It is naturally radioactive and unstable, and will therefore spontaneously decay back into N-14 over a period of time. This process causes a proton to be displaced by a neutron, effectively turning atoms of Nitrogen it into an isotope of carbon – known as”radiocarbon”. Carbon 14 is another, an isotope of carbon that is produced when Nitrogen (N-14) is bombarded by cosmic radiation. C-12, so-named because it has an atomic weight of 12 – is the most common isotope, but it is by no means the only one. Here on Earth, Carbon is found in the atmosphere, the soil, the oceans, and in every living creature.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
